Development of Synthetic Methodology and Approaches to Modified Nucleobases, Nucleosides and Nucleotides
Although our group is primarily not aiming at synthetic methodology development, we need to develop new methodologies and approaches for the synthesis of modified nucleobases, nucleosides and nucleotides for the applications in medicinal and bioorganic chemistry projects.
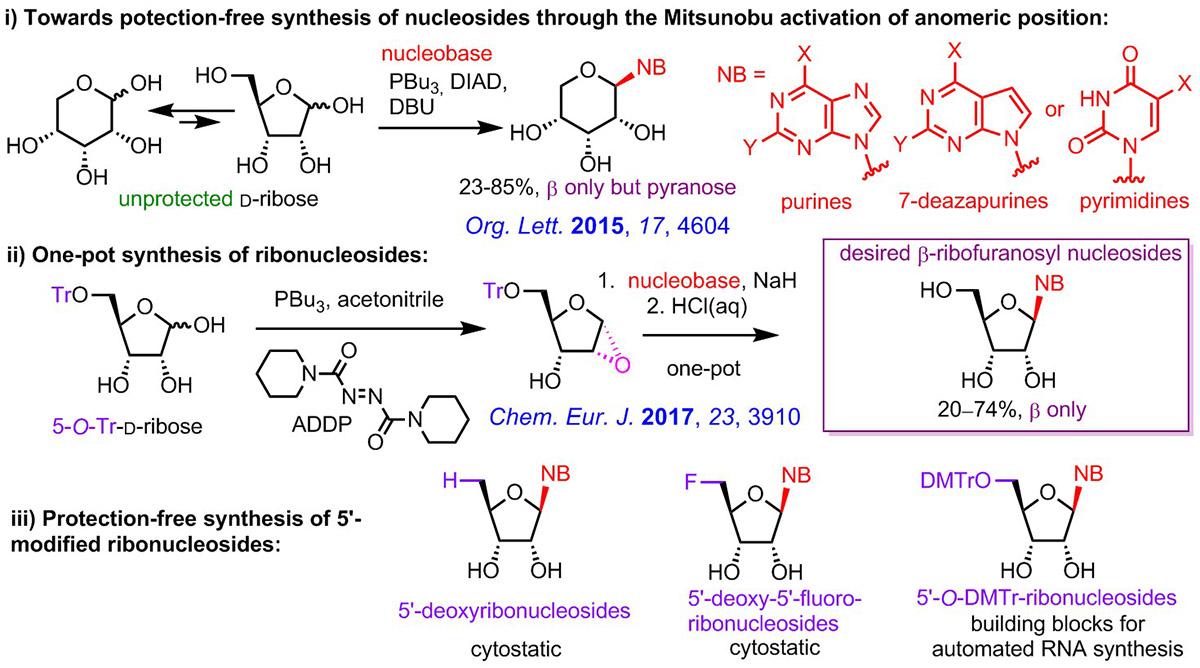
The most important recent synthetic methodology achievement was the discovery of a new glycosylation method for the synthesis of nucleosides. Through optimization of the Mitsunobu reaction of unprotected ribose with nucleobases, we found reagents and conditions allowing to chemo- and stereoselectively attach nucleobase at the anomeric position in the β-configuration but unfortunately in the undesired pyranoside form. However, by fixing the furanose form of the ribose by 5-Tr-protection and using acidic aqueous work-up, the one-pot procedure gave directly the desired β-ribofuranosyl nucleosides. Detailed mechanistic study revealed that the 1,2-epoxide (anhydrose) is formed in situ and reacts with nucleobase. The method is suitable for efficient one-pot synthesis of ribonucleosides derived from purine, 7-deazapurine and pyrimidine nucleobases, as well as for the synthesis of diverse 5'-modified nucleosides (5'-deoxy, 5'-fluoro, 5'-DMTr-protected etc.).
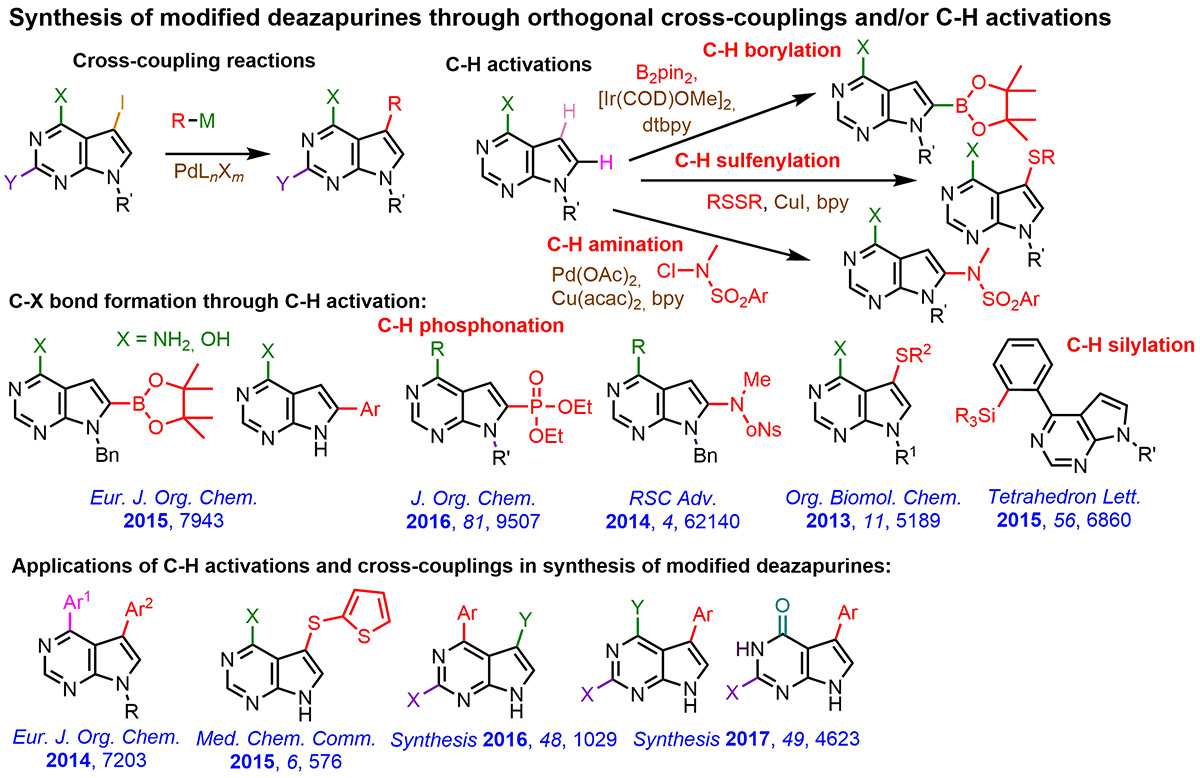
Following our previous achievements in methodology of cross-coupling and C-H activation reactions for modifications of nucleobases, nucleosides and nucleotides, we continued our efforts in developing C-H activation reactions of 7- and 9-deazapurine bases. We focused mostly on the formation of C-X or C-C bonds and we successfully applied C-H borylations, aminations, phosphonations and silylations. In combination with cross-couplings, we synthesized large series of di-, tri- and tetrasubstituted deazapurine bases and tested their biological activity. The screening revealed some novel types of cytostatic molecules which will be further studied and published in the future.